Precision in Every Data Point
Safety in Every Life
A purpose-built 107-bed clinical pharmacology unit in Thaltej, Ahmedabad, delivering high-sensitivity LC-MS/MS bioanalysis and fully compliant BA/BE studies for pharmaceutical sponsors worldwide.

Scientific Excellence
Engineered for Trust
Integrity
Scientific honesty and complete transparency in every study, with zero tolerance for data compromise.
Innovation
Advanced high-sensitivity LC-MS/MS methodologies that enable precise quantification of challenging molecules at sub-picogram levels.
Quality
Independent Quality Assurance with rigorous risk-based audits, full ALCOA+ data integrity, and SOP governance across all phases.
Participant Safety
Continuous 24×7 medical oversight by ACLS/BLS-certified physicians and nursing staff, ensuring the highest level of volunteer safety.
World-Class Infrastructure




















Clinical & Bioanalytical Excellence
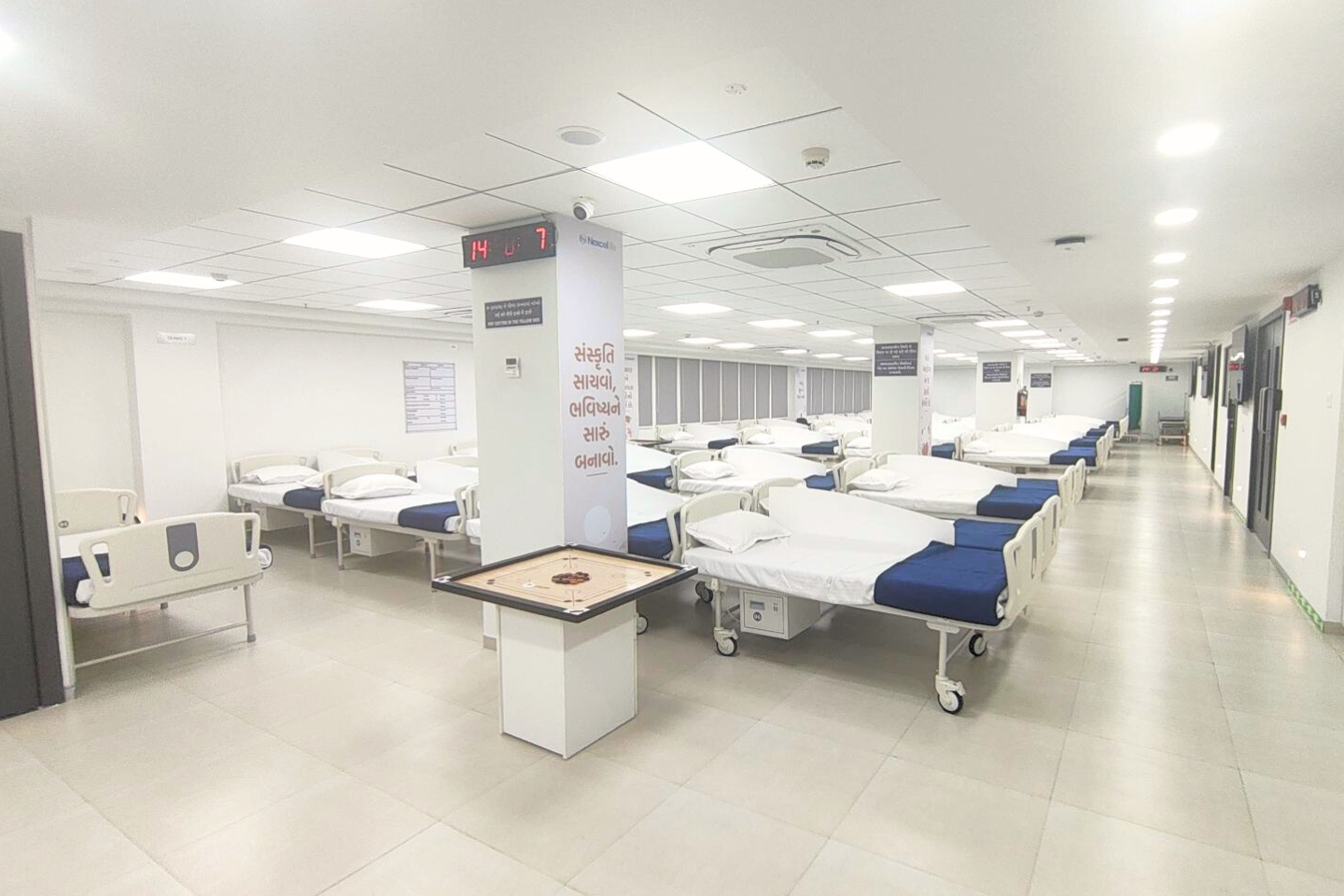





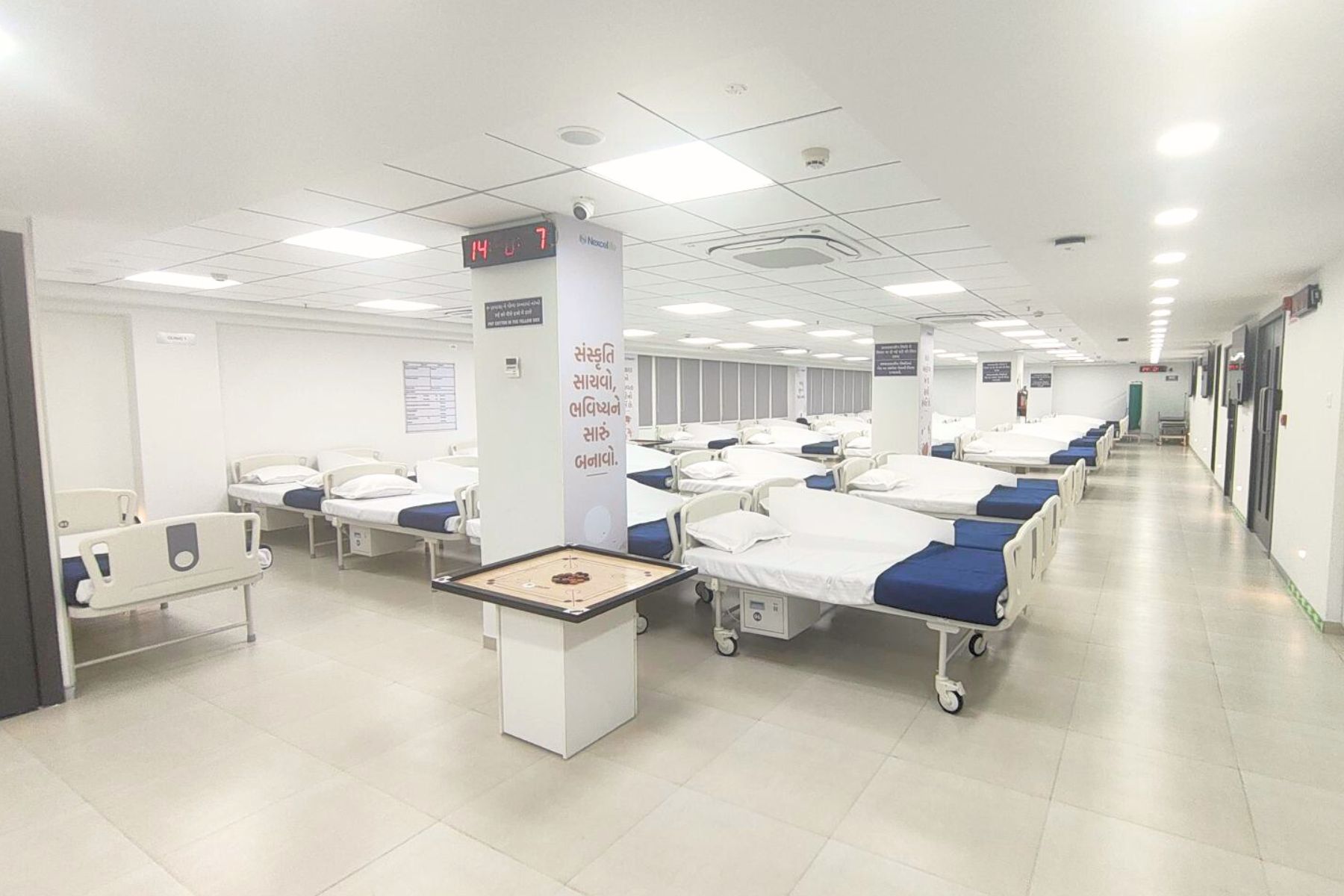
107-Bed Clinical Pharmacology Unit
A modern, single-site CPU approved by the Central Drug Standard Control Organization (CDSCO) for conducting clinical studies, optimized for parallel BA/BE studies without cross-contamination risk.
- Clinic 1: 54 beds | Clinic 2A: 25 beds | Clinic 2B: 28 beds
- 24×7 ACLS-certified medical and nursing supervision
- Biometric access control and dual power backup






Advanced LC-MS/MS Platforms
High-throughput bioanalysis with exceptional sensitivity and reproducibility for complex molecules.
- Triple-quadrupole mass spectrometers supporting MRM analysis
- Validated −70°C storage with continuous temperature monitoring
- Light-protected areas for photosensitive compounds
Integrated Capabilities
Three specialized departments operate in seamless coordination to deliver end-to-end BA/BE services — from protocol design through regulatory submission — with uncompromising quality.
Clinical Pharmacology Unit
107 beds across three segregated clinics, purpose-designed for efficient, contamination-free parallel study execution under 24×7 physician oversight and strict access controls.
CPU · 107 BedsBioanalytical Department
Cutting-edge LC-MS/MS instrumentation delivering sub-pg/mL sensitivity for small molecules, endogenous compounds, and unstable analytes, supported by ICH M10-compliant validation.
LC-MS/MS · ICH M10Quality Assurance
Fully independent QA function enforcing ALCOA+ principles, risk-based monitoring, and rigorous SOP governance to ensure data integrity and zero critical audit findings.
GLP · GCP · ALCOA+Comprehensive BA/BE Services
End-to-end support for global pharmaceutical sponsors seeking reliable, regulator-ready data for generic and complex generic approvals.
Bioequivalence Studies
Healthy-volunteer BA/BE and comparative bioavailability studies conducted in full compliance with CDSCO, USFDA, Health Canada, and EMA requirements, including complex generics and special formulations.
Pharmacokinetic Analysis & Reporting
Expert non-compartmental and compartmental PK modeling using Phoenix WinNonlin, together with comprehensive biostatistics, data management, and eCTD-ready clinical study reports.
Regulatory Submission Support
Full regulatory affairs services, including protocol development, agency liaison, query response management, and preparation of submission dossiers for ANDA, ANDS/NDS, and equivalent pathways.
Build Your Future at India’s Focused BA/BE CRO
We are assembling a team of dedicated scientists, coordinators, and specialists guided by leaders with decades of GLP/GCP experience. Integrity, precision, and continuous professional development define our culture.
CV Received
Thank you. Your CV has been sent to chintan.dholakia@nexcellife.com.

Our Headquarter
Strategically situated in Thaltej, Ahmedabad — within Gujarat’s dynamic life-sciences corridor — with excellent connectivity to major hospitals, research institutions, and international airports.
Bldg on TPS 37,
Opp. Jalaram Kanya Chhatralay,
Altius 3 Road, Thaltej, Ahmedabad,
Gujarat 380059, India